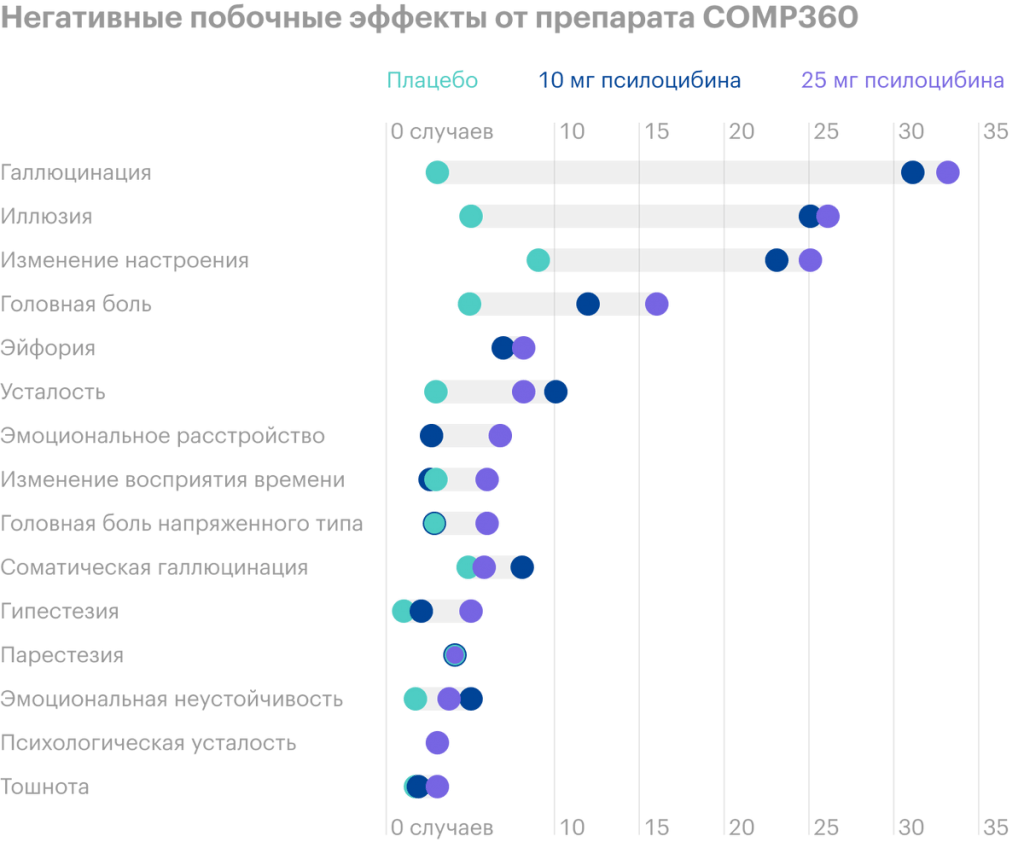
Realizing the futility of treating depressive conditions with traditional drugs using the example of their loved ones, George Goldsmith, Ekaterina Malievskaya and Lars Wild founded Compass Pathways (CP). Synthetic psychedelic psilocybin was chosen as an innovative treatment for depression, found in hallucinogenic mushrooms, - in combination with psychological support. The new drug was named COMP360, and in 2018 US FDA (FDA) approved clinical trials of the drug on volunteers. What's going on here Readers have long asked us to start examining the financial statements and business fundamentals of American issuers. Suggest in the company's comments, analysis of which you would like to read. There are many screenshots with tables from reports in the overview. To make it easier to use them, we translated them into Google tables and translated them into Russian. note: there are several sheets. And keep in mind, that companies round up some numbers in their reports, therefore, the totals in graphs and tables may not converge. Download the table from the report In 2019, CP completed the first phase of drug trials on the group, formed from 89 volunteers. Tests showed good results in almost all subjects, reducing the primary symptoms of the disease within six months after taking the drug. In 2020, a second group of 216 volunteers was formed for the second phase of testing. The second phase of clinical trials is aimed at finding the optimal dosage of COMP360 for various forms of depressive disorder and in combination with the support of a psychotherapist. The company plans to publish the results of the second phase of research in the fourth quarter of 2021. Сравнение рынков и перспективы Производители каннабиса заинтересованы в производстве марихуаны и не занимаются психическим здоровьем своих будущих потребителей. Nevertheless, the hype around this topic artificially warmed up the market, а котировки акций производителей легальной марихуаны …
Обзор Compass Pathways: mushrooms, research and mental health Read more